GNcalc. Software for the calculation of Natural Gas Properties
Introduction
GNcalc is used to calculate the main properties of natural gas such as superior calorific value, inferior calorific value, density, relative density, compressibility factor, molecular weight and Wobbe index. The results depends on the gas composition and the calculations are performed for different reference temperatures and different combustion temperatures.
Run the application
Execute the file GNcalc.exe. The main form will be shown.
Main data
Enter the gas composition:
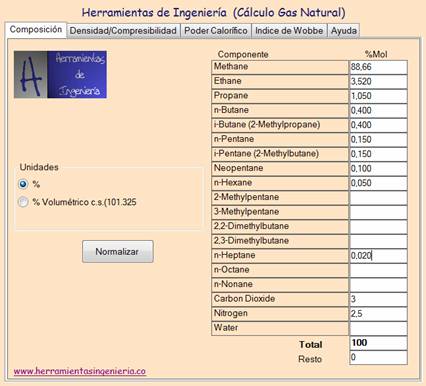
Also select whether the values are expressed in molar percentage or volumetric percentage.
Press the button "Normalize" in order to normalize the composition. This function is used when the sum of all components is different to 100%. In that case the compositions are adjusted so that the sum is exactly 100%. An example is when the composition comes from the output of a chromatograph. In this case analysis will not produce a sum of exactly 100% as it would mean a perferct accuracy of the analyzer. It is a common practice to normalice this output to improve the results.
Once the gas composition has been entered, the rest of tabs will show the results:
·
Density @ 0, 15 y 20??C
·
Relative density @ 0, 15 y 20??C
·
Compressibility @ 0, 15 y 20??C
·
Average molecular weitght
· Superior Calorific Value at different meassurement/combustion temperatures (0/0,15/15,15/0 ºC)
·
Inferior Calorific Value at different meassurement/combustion temperatures (0/0,15/15,15/0 ºC)
·
Higher Wobbe Index at different messurement/combustion temperatures (0/0, 15/15 ºC).
In all cases it is considered the atmospheric pressure (101.325KPa) as specified in the relevant standards.
IMPORTANT CONCEPTS:
Gas Natural Combustion
The combustion of the natural gas consist on a chemical reaction with Oxygen which produce water, CO2 and a big amount of energy. Due to the high temperature that is reached during the combustion the water is in gas phase. Once the combustion gases are cooled down this water will condense, producing an additional amount of energy (vaporization heat of water)
- When in addition to the heat produced from the chemical reaction we take into account also the heat produced by the water condensation, we talk about the Superior Calorific Value.
- If we only consider the heat from the chemical reaction, we are talking about Inferior Calorific Value.
It is important to differentiate between the water that is a product of the combustion.and the water that was already part of the natural gas composition before the combustion has taken place.The water that was already part of the natural gas composition does not affect to the difference between superior and inferior calorific values but it must be also taken into account in the calculations because as calorific value of water is zero, the moisture of natural gas will reduce its calorific value (both superior and inferior).
Natural gas always contains some water which would be desirable to be as low as possible. Frequently the calorific value is considered on a dry basis what means that it is assumed that the gas contains no water vapor.The chromatographs used for calorific value determination must work with dry samples so that the moisture is removed from de sample before being analyzed, so the output from chromatographs are on dry basis (although they can also calculate some properties for saturated gas). On the other hand we have the calorific value for water saturated natural gas.
The reference standard for the calculation of calorific values from composition is ISO 6976. This standard also describes the procedure to calculate density, relative density and Wobbe index
Superior calorific value:
Is the amount of heat which would be released by the complete combustion in air of a gas, and all the products of combustion are returned to the same specified temperature t1 as that of the reactants, all of these products being in the gaseous state except for water formed by combustion, which is condensed to the liquid state at t1.
Inferior calorific value:
Is the amount of heat which would be released by the complete combustion in air of a gas and all the product of combustion are returned to the same specifed tmperature t1 as that of the reactants, all of these products being in the gaseous state.
Density
The mass of a gas divided by its volume at specified conditions of pressure and temperature.
Relative density:
The density of a gas divided by the density of dry air at the same pressure and temperature.
Wobbe index
The superior calorific value on a volumetric basis, divided by the square root of the relative density.
The Wobbe index is a indicator of the intechambiability of gas combustibles. Two different gases with the same Wobbe index, same pressure and feeding the same burner will release the same quantity of energy. As the relative density is adimensional, Wobbe index units and calorific value have the same units.
Compression factor (Z)
The actual volume of a given mass of gas divided by its volume, under the same conditions, as calculated from de ideal gas law. It is a indication of how a real gas behaviour differs from a ideal gas. For ideal gases Z=1..
The ideal gas law ( P·V = n · R · T) is corrected with this factor, as follows:
P·V = Z · n · R · T
Combustion reference conditions:
Pressure and temperaure at which the fuel is burned.
Metering reference conditions:
Temperature and pressure at which the amount of fuel to be burned is determined.